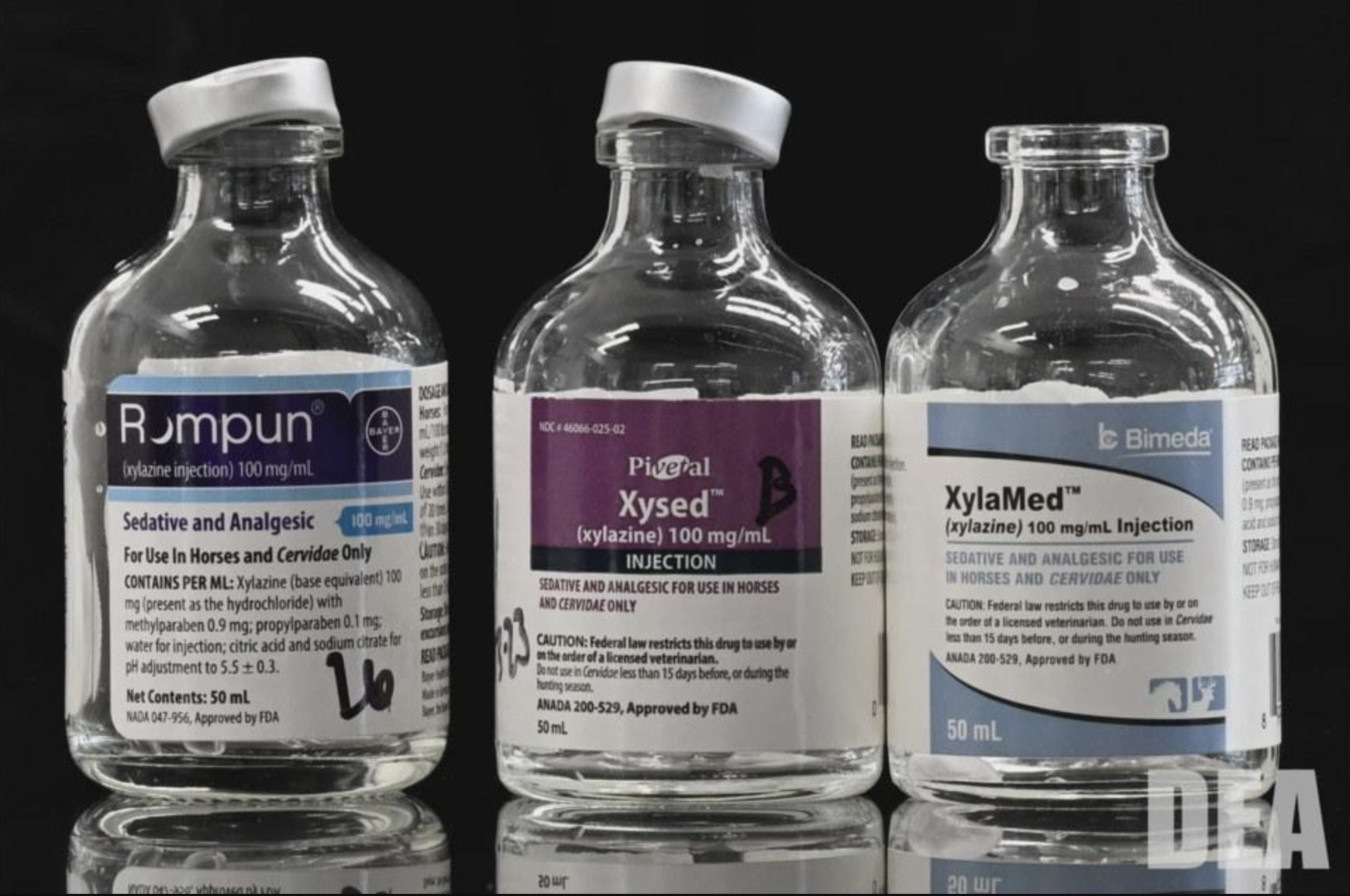
Three years in the making, the legislative push to place xylazine under Schedule III of the federal Controlled Substances Act is nearing the finish line.
On March 26, the Combatting Illicit Xylazine Act was advanced by the Senate Judiciary Committee with a vote of 19-3, setting the stage for a vote before the full chamber. In a House Committee on Energy and Commerce hearing the same day, proponents warned about how dangerous it would be if Congress didn’t schedule xylazine—not because xylazine would remain unscheduled, but because the DEA would do it instead.
Xylazine is a non-opioid sedative that’s been approved by the Food and Drug Administration as a veterinary tranquilizer for more than 50 years. It was starting to become prevalent in the East Coast unregulated opioid supply just before the COVID-19 pandemic, during which its role expanded significantly. Fentanyl doesn’t last as long as heroin, leaving people in a cycle of using more often and getting sick again just as quickly, and so xylazine began to show up mixed in to stretch the fentanyl and give it “legs.”
Those who were lucky were able to get the harm reduction benefits, like injecting less frequently or having less exposure to law enforcement. But xylazine also introduced an array of serious harms—protracted blackouts; painful withdrawal; overdose that was more difficult to reverse; soft-tissue infections that turned necrotic and seemed to never heal.
The Combatting Illicit Xylazine Act is a reintroduction of a version first proposed in March 2023, shortly before the Biden administration designated fentanyl-xylazine mixture an “emerging threat”—the first time that the White House had applied that designation to the drug supply. As a Schedule III substance xylazine would become somewhat difficult to access, but the Act contains an “ultimate user” carveout for veterinarians and pharmacists, as well as others who could demonstrate that they own or work with livestock—farmers, ranchers and so forth. Legislation, however, is one of two ways that a substance can be federally scheduled. The DEA can do it through a separate process, which has also been underway since 2023. It doesn’t give the agency the authority to write in any exemptions.
In addition to both the Biden and Trump administrations, the bill has been endorsed by the veterinary medical associations of all 50 states.
“Absent congressional action, our understanding is that the DEA will administratively schedule xylazine,” Fred Gingrich, executive director of the American Association of Bovine Practitioners, stated during the March 26 hearing. “[This] would have profound consequences. Veterinarians could lose access to this critical medication with no viable substitute, increasing risk to practitioners, animal handlers and the public. Animal welfare would be compromised and the ability to provide timely and humane care would be diminished.”
Gingrich, who testified on behalf of the American Veterinary Medical Association (AVMA), emphasized at several points that xylazine is the only viable option for sedating cattle. The AVMA has lobbied heavily for the bill.
Schedule III is ostensibly for drugs that have a “moderate to low potential for physical and psychological dependence.” This category includes ketamine, testosterone and buprenorphine (Suboxone). However, CSA scheduling is notoriously inconsistent. For example, benzodiazepines, which the DEA characterizes as having a “high potential for abuse,” are mostly Schedule IV, while marijuana has yet to be moved from Schedule I, the category for drugs with no recognized medical benefit.
Recent research indicates that xylazine does in fact respond to naloxone.
A fact sheet for the legislation claims that xylazine “is increasingly the cause of fatal overdoses, in part because it’s resistant to Narcan.”
This is wrong from every possible angle. Xylazine’s presence in the supply has been rapidly dwindling since early 2025, and in its place we have medetomidine—a different veterinary sedative.
“The wrong schedule can make a bad decision worse,” Dr. Nabarun Dasgupta, who runs the Opioid Data Lab and public drug-checking program at the University of North Carolina at Chapel Hill, testified in opposition to the bill. “In Pittsburgh, hospitals are filling up not with overdose, but with people who try to stop using what they thought was fentanyl but had medetomidine. This unintended consequence can be traced directly back to the moment when Pennsylvania permanently put xylazine in Schedule III … the natural thought might be to schedule both xylazine and medetomidine, but neither of these drugs really belongs in Schedule III, scientifically.”
Though lawmakers, law enforcement and media use this phrase fairly regularly, non-opioid drugs like xylazine aren’t “resistant” to naloxone (Narcan); naloxone was not designed nor expected to ever have an effect on them in the first place. But as xylazine is overwhelmingly found in conjunction with fentanyl, the best advice, including from the Centers for Disease Control and Prevention, has been to always administer naloxone, and extended rescue breathing if needed.
However, contrary to popular belief, recent research at UNC indicates that xylazine does in fact respond to naloxone. Dasgupta stated during the hearing that naloxone acts directly on xylazine, both alone and in combination with fentanyl. Filter confirmed that this refers to overdose reversal, and not other contexts like withdrawal symptoms. Naloxone doesn’t act on the opioids themselves, but rather the receptors in our systems that the opioid molecules bind to; xylazine binds to some of those same receptors as well.
“It doesn’t wake people up and animate people,” Dasgupta continued. “So what we say is carry naloxone, administer it, but count breaths and you’ll see that the breathing comes back. But they may not get up and moving for a couple hours, you want to stay with them so that they don’t get raped or have their stuff stolen.”
Image (cropped) via United States Drug Enforcement Administration