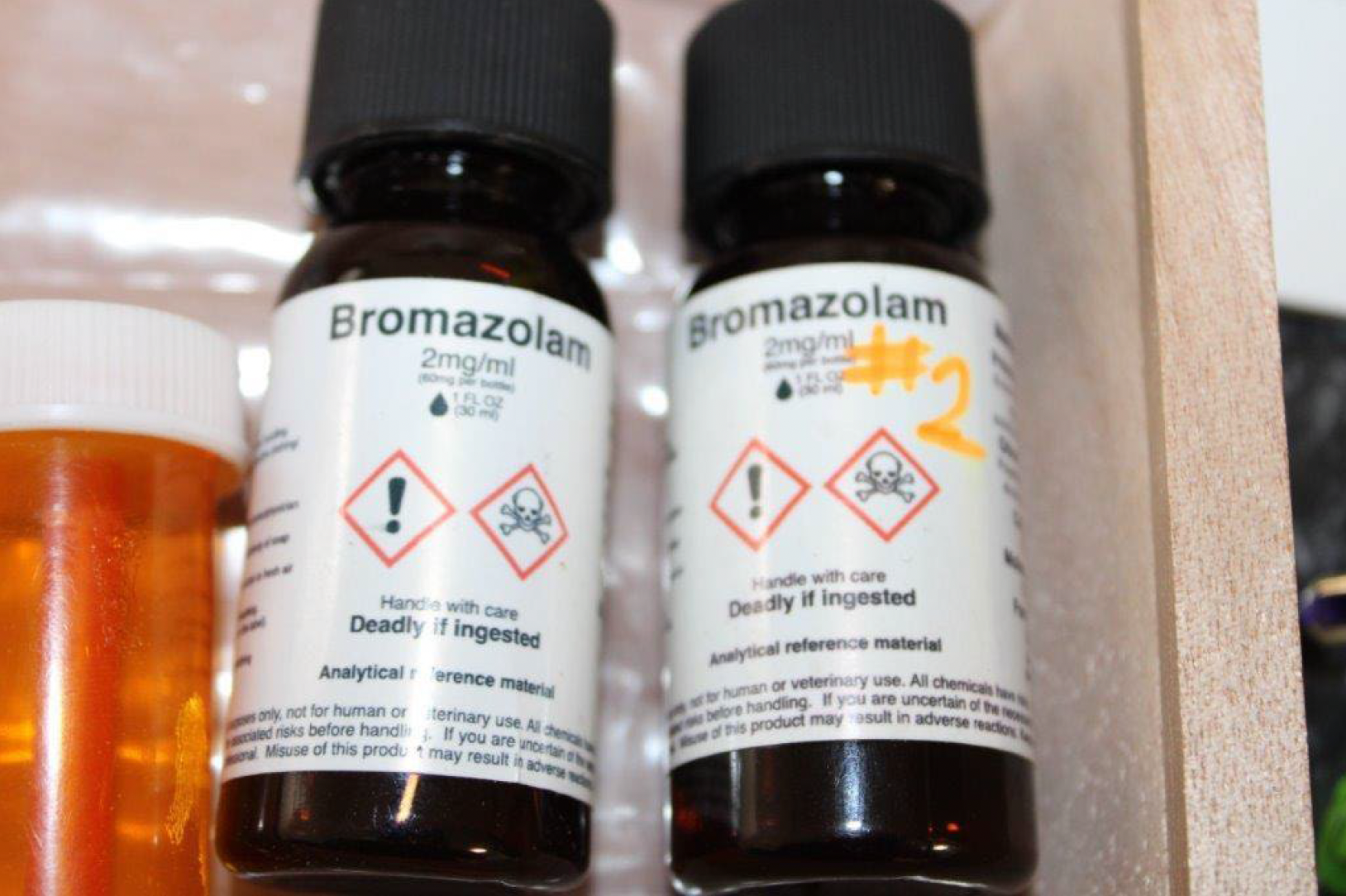
The Drug Enforcement Administration has temporarily placed bromazolam, an unregulated benzodiazepine, under Schedule I of the Controlled Substances Act. The emergency order was issued March 16 and will remain in effect until the same date in 2028. The DEA has been escalating its temporary and permanent scheduling of new synthetic drugs, which is exactly the type of enforcement that results in newer synthetic drugs.
Bromazolam (which the DEA formally identifies as 8-bromo-1-methyl-6-phenyl-4 H -benzo[ f][1,2,4]triazolo[4,3- a][l, 4]diazepine) is similar to alprazolam (Xanax). It’s often found in the counterfeit pressed pills that are designed to look like Xanax, but can show up in powder fentanyl or in pressed pills designed to look like oxycodone or other opioids. It’s mainly associated with fentanyl, but frequently identified alongside other substances including meth and cocaine, and other novel synthetic drugs like nitazenes that also tend to be associated with fentanyl.
For the past several years, bromazolam has been the most prevalent unregulated benzo in the United States drug supply. It’s central to what many in the US know as “benzo dope,” though elsewhere this has been more associated with other benzos like etilozam. It was first encountered by US law enforcement in 2016, but it didn’t emerge as particularly significant until around late 2022, when etilozam—the unregulated benzo that had been most prevalent at that time—suddenly dropped out of the supply, and bromazolam surged in its place.
Our 2024 NPS Discovery Trend Report shows novel benzodiazepines account for 50% of NPS identifications in the U.S. Fatal overdoses often involve these and opioids. Etizolam and Flualprazolam declined since 2022, while Bromazolam’s prevalence has increased. pic.twitter.com/iIcKLZg6Zc
— CFSRE (@CFSRE_) October 10, 2024
The push to add bromazolam to the CSA was initiated in 2024 under the Biden administration, but the intention to order temporary scheduling was formally announced in December 2025. In its new temporary order, the DEA reported that bromazolam was involved in at least 201 overdose deaths between April 2021 and February 2026. However, the vast majority of these also involved fentanyl; only four deaths were attributed to bromazolam alone. The DEA stated that “[a]dditional sources of information demonstrate additional overdoses, which would suggest that this statistic is likely subject to underreporting.”
In the fourth quarter of 2025, the Center for Forensic Science Research & Education identified bromazolam in 145 toxicology samples (which could include samples from post-mortem examinations, but also from people who were alive.) The next most prevalent benzo, desalkylgidazepam, was identified in just 39 toxicology samples and zero seized drug samples.
Though Schedule I is often framed as the category for the drugs that are the most dangerous, almost all of the most heavily stigmatized and surveilled drugs that the public is familiar with—fentanyl, methamphetamine, cocaine—are Schedule II, because the Food and Drug Administration has approved them for one or more medical purposes. (Heroin, which is not approved for any medical use in the US, is Schedule I.)
Schedule I is increasingly populated with novel synthetic drugs that the public hasn’t heard of, and which play a relatively minor role in the drug supply. Unlike more established drugs, they have no existing body of research to stop the DEA from whisking them away into the CSA’s most restrictive category. This is why fentanyl remains Schedule II, but fentanyl analogs that have only been identified a handful of times in comparison are under Schedule I.
Earlier in March, the DEA issued a permanent order placing clonazolam, diclazepam, etizolam, flualprazolam and flubromazolam in Schedule I—the first benzos to be permanently scheduled in that category. All five had been temporarily scheduled for several years.
In January, the DEA issued a temporary scheduling order for a ketamine analog, but in recent years it has focused such orders on synthetic cannabinoids, nitazenes and fentanyl analogs.
Temporary scheduling is never a sign that the DEA isn’t fully convinced that a substance belongs in the CSA. It’s the step that precedes permanent scheduling, or sometimes just a way for the DEA to bypass inconvenient democratic safeguards.
“Protecting our communities from emerging drugs is a top priority,” DEA Diversion Control Division Assistant Administrator Cheri Oz stated March 18, “and we will continue to act swiftly to stay ahead of those who seek to exploit them.”
Images (cropped) via Regulations.gov/Drug Enforcement Administration